Ulcerative colitis is a chronic inflammatory and ulcerative disease arising in the colonic mucosa, characterized most often by bloody diarrhea. Extraintestinal symptoms, particularly arthritis, may occur. Long-term risk of colon cancer is elevated compared to unaffected people. Diagnosis is by colonoscopy. Treatment is with 5-aminosalicylic acid, glucocorticoids, biologics, immunomodulators, small molecules, and occasionally surgery.
(See also Overview of Inflammatory Bowel Disease.)
Pathophysiology of Ulcerative Colitis
Ulcerative colitis usually begins in the rectum. It may remain localized to the rectum (ulcerative proctitis) or extend proximally, sometimes involving the entire colon. Rarely, it involves most of the large bowel at once.
The inflammation caused by ulcerative colitis affects the mucosa and submucosa, and there is a sharp border between normal and affected tissue. Only in severe disease is the muscularis involved. Early in the disease, the mucous membrane is erythematous, finely granular, and friable, with loss of the normal vascular pattern and often with scattered hemorrhagic areas. Large mucosal ulcers with copious purulent exudate characterize severe disease. Islands of relatively normal or hyperplastic inflammatory mucosa (pseudopolyps) project above areas of ulcerated mucosa. Fistulas and abscesses do not occur (as they do in Crohn disease).
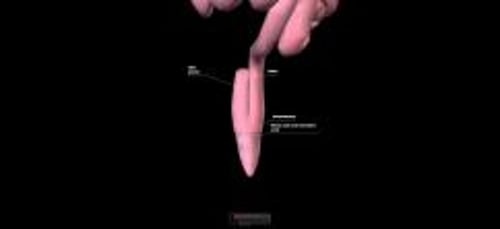
The colonoscopic findings in ulcerative colitis shown here include edema, loss of vascularity, friable mucosa, and ulcers.
Pseudopolyps are areas of hyperplastic, inflamed mucosa that project above the mucosal surface.
Toxic colitis
Toxic colitis or fulminant colitis occurs when transmural extension of ulceration results in localized ileus and peritonitis. Within hours to days, the colon loses muscular tone and begins to dilate.
The terms toxic megacolon and toxic dilation are discouraged because the toxic inflammatory state and its complications can occur without frank megacolon (defined as transverse colon > 6 cm diameter during an exacerbation).
Toxic colitis is a medical emergency that usually occurs spontaneously in the course of very severe colitis but is sometimes precipitated by opioid or anticholinergic antidiarrheal medications. Colonic perforation may occur, which increases mortality significantly.
Symptoms and Signs of Ulcerative Colitis
Patients typically have
Attacks of bloody diarrhea of varied intensity and duration interspersed with asymptomatic intervals
Usually an attack begins insidiously, with increased urgency to defecate, mild lower abdominal cramps, and blood and mucus in the stools. Some cases develop after an infection (eg, amebiasis, bacillary dysentery).
When ulceration is confined to the rectosigmoid, the stool may be normal or hard and dry, but rectal discharges of mucus loaded with red and white blood cells accompany or occur between bowel movements. Systemic symptoms are absent or mild.
If ulceration extends proximally, stools become looser and the patient may have > 10 bowel movements per day, often with severe cramps and distressing rectal tenesmus, without respite at night. The stools may be watery or contain mucus and frequently consist almost entirely of blood and pus.
Toxic or fulminant colitis manifests initially with sudden violent diarrhea, fever to 40° C (104° F), abdominal pain, signs of peritonitis (eg, rebound tenderness), and profound toxemia.
Systemic symptoms and signs, more common with extensive ulcerative colitis, include malaise, fever, anemia, anorexia, and weight loss.
Extraintestinal manifestations of inflammatory bowel disease (IBD), particularly joint and skin complications, are most common when systemic symptoms are present. Extraintestinal manifestations seen during flares of gastrointestinal disease include arthritis, episcleritis, and erythema nodosum. Other extraintestinal symptoms, such as pyoderma gangrenosum, sacroiliitis, and uveitis, may occur in the absence of a gastrointestinal disease flare. The hepatobiliary system may also be involved.
Diagnosis of Ulcerative Colitis
Stool cultures and microscopy (to exclude infectious causes)
Colonoscopy or sigmoidoscopy with biopsy
Laboratory evaluation
Sometimes radiography
Initial evaluation and differential diagnosis
Diagnosis of ulcerative colitis is suggested by typical symptoms and signs, particularly when accompanied by extraintestinal manifestations or a history of previous similar attacks. Ulcerative colitis should be distinguished from Crohn disease (see table ) but more importantly from other causes of acute colitis (eg, infection; in older patients, ischemia).
Diagnostic evaluation should exclude infectious causes, and should include stool assay for Clostridioides difficile (formerly Clostridium difficile) toxin in all patients suspected of having ulcerative colitis (1). Stool cultures for enteric pathogens should be performed, and Entamoeba histolytica should be excluded by examination of fresh stool specimens. When amebiasis is suspected because of epidemiologic or travel history, serologic titers and biopsies should be performed. History of prior antibiotic use or recent hospitalization should prompt testing of at-risk patients for HIV, gonorrhea, herpesvirus, chlamydia, and amebiasis. Opportunistic infections (eg, cytomegalovirus, Mycobacterium avium-intracellulare) or Kaposi sarcoma must also be considered in immunosuppressed patients.
Endoscopy allows visual confirmation of colitis and permits direct sampling of stool or mucus for culture and microscopic evaluation, as well as biopsy of affected areas. Although visual inspection and biopsies may be nondiagnostic, because there is much overlap in appearance among different types of colitis, acute, self-limited, infectious colitis can usually be distinguished histologically from chronic idiopathic ulcerative colitis or Crohn colitis. Severe perianal disease, rectal sparing, absence of bleeding, and asymmetric or segmental involvement of the colon indicate Crohn disease rather than ulcerative colitis. When safe, colonoscopy with examination of the terminal ileum should be performed by an experienced operator (1–3). Flexible sigmoidoscopy with biopsies is an alternative for patients with severe disease and increased risk of perforation.
Laboratory tests should be performed to screen for anemia, hypoalbuminemia, electrolyte abnormalities, systemic inflammation (eg, erythrocyte sedimentation rate, C-reactive protein), thrombocytosis, and leukocytosis (1, 3). The presence of anemia, elevated inflammatory markers, and hypoalbuminemia may be useful for risk stratification. Liver tests should be performed; elevated alkaline phosphatase and gamma-glutamyl transpeptidase levels suggest possible primary sclerosing cholangitis.
Fecal calprotectin is used primarily as a marker of disease activity and treatment response (1–3).
Serologic tests such as perinuclear antineutrophil cytoplasmic antibodies and anti–Saccharomyces cerevisiae antibodies do not reliably differentiate ulcerative colitis from Crohn disease and are not recommended for routine diagnosis (1).
Ischemic colitis should be considered as an alternative diagnosis in older patients. In women using oral contraceptives, contraceptive-induced colitis is possible; it usually resolves spontaneously after hormone therapy is stopped. Stool testing for lactoferrin and fecal calprotectin can be beneficial in differentiating IBD from functional diarrhea.
Immune-mediated colitis (IMC), an immune-related adverse effect associated with immune checkpoint inhibitors, occurs 5 to 10 weeks after the second or third dose of medication. Because immune-mediated colitis is indistinguishable from inflammatory bowel disease and has remarkable overlap in histology, it is important to recognize based on clinical history (4). Optimal management of IMC requires early recognition and timely treatment with glucocorticoids, infliximab, or vedolizumab.). Optimal management of IMC requires early recognition and timely treatment with glucocorticoids, infliximab, or vedolizumab.
Radiographs are not diagnostic but occasionally show abnormalities. Plain radiographs of the abdomen may show mucosal edema, loss of haustration, and absence of formed stool in the diseased bowel; however, the yield is low and they are not recommended routinely. Barium enema shows similar changes, albeit more clearly, and may also show ulcerations, but the enema should not be performed during an acute presentation. A shortened, rigid colon with an atrophic or pseudopolypoid mucosa is often seen after several years of illness. Radiographic findings of thumbprinting and segmental distribution are more suggestive of intestinal ischemia or possibly Crohn colitis rather than of ulcerative colitis.
Assessing disease severity
Disease-activity monitoring in ulcerative colitis combines symptom assessment with biomarkers (eg, fecal calprotectin and C-reactive protein) and selective endoscopic evaluation.
Multiple scoring systems are also available to evaluate disease severity. The Mayo clinical and endoscopic scoring system uses stool frequency, amount of blood in stool, a physician global assessment, and endoscopic appearance to classify disease into mild, moderate, and severe (5). Other assessment tools focus on endoscopic appearance or disease location (1, 3). Poor prognostic factors that may guide the decision to escalate therapy include age <40, extensive disease or severe endoscopic disease activity, hypoalbuminemia, steroid dependence, and elevated C-reactive protein.
Recurrent symptoms
Patients with known disease and a recurrence of typical symptoms should be examined, but extensive testing is not always required. Depending on duration and severity of symptoms, sigmoidoscopy or colonoscopy may be performed and a complete blood count obtained. Cultures, ova and parasite examination, and C. difficile toxin assay should be performed when there are atypical features to the relapse or when there is an exacerbation after prolonged remission, during a contagious outbreak, after antibiotic exposure, or whenever the clinician is suspicious.
Acute severe attacks
Acute severe ulcerative colitis is characterized by more than 6 bloody stools per day with fever, tachycardia, anemia, and elevated inflammatory markers (2).
Patients require prompt hospitalization during severe flares. Flat and upright abdominal radiographs should be taken; they may show megacolon or intraluminal gas accumulated over a long, continuous, paralyzed segment of colon—a result of lost muscle tone. Barium enema should be avoided because of the risk of perforation, but a careful sigmoidoscopy is typically advisable to assess severity and exclude infection. Complete blood count, platelet count, erythrocyte sedimentation rate, C-reactive protein, electrolytes, albumin, and fecal calprotectin should be obtained; prothrombin time, partial thromboplastin time, and blood type and cross-match are also indicated in cases of severe bleeding.
The patient must be watched closely for progressive peritonitis or perforation. Percussion over the liver is important because loss of hepatic dullness may be the first clinical sign of free perforation, especially in a patient whose peritoneal signs are suppressed by high-dose glucocorticoids. Abdominal radiographs are taken every 1 or 2 days to follow the course of colonic distention and to detect free or intramural air; CT is more sensitive in detecting extraluminal air or pericolic abscess.
Diagnosis references
1. Rubin DT, Ananthakrishnan AN, Siegel CA, Barnes EL, Long MD. ACG Clinical Guideline Update: Ulcerative Colitis in Adults. Am J Gastroenterol. 2025;120(6):1187-1224. Published 2025 Jun 3. doi:10.14309/ajg.0000000000003463
2. Gros B, Kaplan GG. Ulcerative Colitis in Adults: A Review. JAMA. 2023;330(10):951-965. doi:10.1001/jama.2023.15389
3. Le Berre C, Honap S, Peyrin-Biroulet L. Ulcerative colitis. Lancet. 2023;402(10401):571-584. doi:10.1016/S0140-6736(23)00966-2
4. Som A, Mandaliya R, Alsaadi D, et al. Immune checkpoint inhibitor-induced colitis: A comprehensive review. World J Clin Cases. 2019;7(4):405-418. doi:10.12998/wjcc.v7.i4.405
5. Schroeder KW, Tremaine WJ, Ilstrup DM. Coated oral 5-aminosalicylic acid therapy for mildly to moderately active ulcerative colitis. A randomized study. N Engl J Med. 1987;317(26):1625-1629. doi:10.1056/NEJM198712243172603
Treatment of Ulcerative Colitis
Dietary management and loperamide (except in acute severe attacks) for symptom reliefDietary management and loperamide (except in acute severe attacks) for symptom relief
Rectal and/or oral 5-aminosalicylic acid (5-ASA) for mild disease
Glucocorticoids, biologic agents, or small molecules for moderate to severe disease
Sometimes surgery
The goal of therapy in patients with ulcerative colitis is to achieve remission and prevent disease complications (1). The treatment approach depends on disease severity and specific location of colonic involvement. Mild to moderate ulcerative proctitis (inflammation limited to < 15 to 20 cm from the anus) is usually treated with rectal 5-ASA (mesalamine) therapies. For patients with left-sided or extensive colitis (inflammation extending proximal to the rectum), combination therapy with oral mesalamine and rectal mesalamine enemas is typically used. Patients with mild to moderate disease who do not respond to mesalamine can also receive a short course of systemic glucocorticoids (eg, budesonide, prednisone). Patients with moderate to severe disease may receive systemic glucocorticoids for induction followed by thiopurines for maintenance therapy; or nonsteroid agents for both induction and maintenance ((mesalamine) therapies. For patients with left-sided or extensive colitis (inflammation extending proximal to the rectum), combination therapy with oral mesalamine and rectal mesalamine enemas is typically used. Patients with mild to moderate disease who do not respond to mesalamine can also receive a short course of systemic glucocorticoids (eg, budesonide, prednisone). Patients with moderate to severe disease may receive systemic glucocorticoids for induction followed by thiopurines for maintenance therapy; or nonsteroid agents for both induction and maintenance (1). Surgery may be required to treat disease complications (eg, strictures, abscesses) or to treat disease refractory to medications.
Details of specific medications and dosages are discussed in Medications for Inflammatory Bowel Disease.
General management
Avoiding raw fruits and vegetables limits trauma to the inflamed colonic mucosa and may lessen symptoms. A milk-free diet may help but need not be continued if no benefit is noted. Loperamide 2 mg orally 2 to 4 times a day is indicated for relatively mild diarrhea; higher oral doses (4 mg in the morning and 2 mg after each bowel movement) may be required for more intense diarrhea. Antidiarrheal medications should be avoided or used with extreme caution in severe cases because they may precipitate toxic dilation. All patients with inflammatory bowel disease should be advised to take appropriate amounts of calcium and vitamin D.Avoiding raw fruits and vegetables limits trauma to the inflamed colonic mucosa and may lessen symptoms. A milk-free diet may help but need not be continued if no benefit is noted. Loperamide 2 mg orally 2 to 4 times a day is indicated for relatively mild diarrhea; higher oral doses (4 mg in the morning and 2 mg after each bowel movement) may be required for more intense diarrhea. Antidiarrheal medications should be avoided or used with extreme caution in severe cases because they may precipitate toxic dilation. All patients with inflammatory bowel disease should be advised to take appropriate amounts of calcium and vitamin D.
Routine health maintenance measures (eg, immunizations, cancer screening) should be emphasized (2). In general, recommended health maintenance schedules should be followed. Prior to treatment with immunomodulators or biologic therapy, patients should receive all recommended inactivated vaccines and be screened for latent tuberculosis, hepatitis B, and hepatitis C. Live virus vaccines should be avoided in immunosuppressed patients.
Mild left-sided disease
Patients with mild to moderate ulcerative proctitis or proctosigmoiditis that does not extend proximally beyond the sigmoid colon are treated with 5-ASA (mesalamine) by enema once or twice a day depending on severity ((mesalamine) by enema once or twice a day depending on severity (1, 3). Suppositories are effective for more distal disease and are usually preferred by patients. Oral 5-ASA medication is added for left-sided disease, or for proctitis not responsive to topical therapy. Glucocorticoid and budesonide enemas are slightly less effective but can be used if 5-ASA is unsuccessful or not tolerated. Tacrolimus suppository or beclomethasone suppository can also be used for patients with mildly to moderately active ulcerative proctitis not responsive to topical 5-ASA. Budesonide MMX 9 mg/d can also be used for induction of remission in mild to moderately active left-sided disease. Once remission is achieved, dosage is slowly tapered to maintenance levels.). Suppositories are effective for more distal disease and are usually preferred by patients. Oral 5-ASA medication is added for left-sided disease, or for proctitis not responsive to topical therapy. Glucocorticoid and budesonide enemas are slightly less effective but can be used if 5-ASA is unsuccessful or not tolerated. Tacrolimus suppository or beclomethasone suppository can also be used for patients with mildly to moderately active ulcerative proctitis not responsive to topical 5-ASA. Budesonide MMX 9 mg/d can also be used for induction of remission in mild to moderately active left-sided disease. Once remission is achieved, dosage is slowly tapered to maintenance levels.
Moderate or extensive disease
Patients with inflammation proximal to the sigmoid colon or left-sided disease unresponsive to topical agents should receive a biologic agent (infliximab, adalimumab, golimumab, guselkumab, risankizumab, mirikizumab, ustekinumab, vedolizumab) with or without an immunomodulator medication (azathioprine, methotrexate, 6-mercaptopurine) ((infliximab, adalimumab, golimumab, guselkumab, risankizumab, mirikizumab, ustekinumab, vedolizumab) with or without an immunomodulator medication (azathioprine, methotrexate, 6-mercaptopurine) (1, 4, 5). Sometimes, high-dose glucocorticoids are added for inducing remission in patients with moderate to severe ulcerative colitis. Finally, in some patients, a trial of the small-molecule Janus kinase inhibitors (tofacitinib or upadacitinib) or the sphingosine 1-phosphate receptor modulator ozanimod or etrasimod can be considered. Methotrexate or thiopurine monotherapy are not recommended.are added for inducing remission in patients with moderate to severe ulcerative colitis. Finally, in some patients, a trial of the small-molecule Janus kinase inhibitors (tofacitinib or upadacitinib) or the sphingosine 1-phosphate receptor modulator ozanimod or etrasimod can be considered. Methotrexate or thiopurine monotherapy are not recommended.
Severe disease
Patients with > 6 bloody bowel movements per day, tachycardia, high fever, or severe abdominal pain require hospitalization to receive high-dose IV glucocorticoids and/or a biologic agent (1, 4, 5). C difficile and CMV colitis should first be excluded or treated if found. Deep venous thrombosis prophylaxis should be given. IV fluids and blood transfusion are given as needed for dehydration and anemia. The patient must be observed closely for the development of toxic colitis. Parenteral hyperalimentation is sometimes used for nutritional support but is of no value as primary therapy; patients who can tolerate food should eat.
Patients who do not respond to biologic agents and/or glucocorticoids within 3 to 7 days should be considered for IV cyclosporine or for surgery (1). Patients who do respond to a glucocorticoid regimen are switched within a week or so to prednisone 40 mg orally once a day, which may be gradually reduced at home based on clinical response. Patients who are started on IV cyclosporine and respond to therapy are switched to oral cyclosporine and concomitant azathioprine or 6-mercaptopurine. Oral cyclosporine is continued for about 3 to 4 months, during which time glucocorticoids are tapered and cyclosporine levels are closely monitored. Some clinicians recommend ). Patients who do respond to a glucocorticoid regimen are switched within a week or so to prednisone 40 mg orally once a day, which may be gradually reduced at home based on clinical response. Patients who are started on IV cyclosporine and respond to therapy are switched to oral cyclosporine and concomitant azathioprine or 6-mercaptopurine. Oral cyclosporine is continued for about 3 to 4 months, during which time glucocorticoids are tapered and cyclosporine levels are closely monitored. Some clinicians recommendprophylaxis against Pneumocystis jirovecii pneumonia during the interval of overlapping treatment with glucocorticoids, cyclosporine, and an antimetabolite. Tacrolimus, an immunosuppressant also used in transplant patients, seems as effective as cyclosporine and may be considered for use in patients with severe or refractory ulcerative colitis who do not require hospitalization.pneumonia during the interval of overlapping treatment with glucocorticoids, cyclosporine, and an antimetabolite. Tacrolimus, an immunosuppressant also used in transplant patients, seems as effective as cyclosporine and may be considered for use in patients with severe or refractory ulcerative colitis who do not require hospitalization.
Fulminant colitis
If fulminant or toxic colitis is suspected (>10 stools per day with continuous blood, colonic dilation with fever, tachycardia, anemia, and elevated inflammatory markers) (6), the patient should
Stop all antidiarrheal medications
Take nothing by mouth and have inserted a long intestinal tube attached to intermittent suction
Receive aggressive IV fluid and electrolyte therapy with isotonic saline, with potassium and blood as needed
Receive high-dose IV glucocorticoids or cyclosporineReceive high-dose IV glucocorticoids or cyclosporine
Receive antibiotics (eg, metronidazole 500 mg IV every 6 to 8 hours and ciprofloxacin 500 mg IV every 12 hours)Receive antibiotics (eg, metronidazole 500 mg IV every 6 to 8 hours and ciprofloxacin 500 mg IV every 12 hours)
Perhaps receive infliximabPerhaps receive infliximab
Having the patient roll over in bed from the supine to prone position every 2 to 3 hours may help redistribute colonic gas and prevent progressive distention. Insertion of a soft rectal tube may also be helpful but must be performed with extreme caution to avoid bowel perforation. Even if decompression of a dilated colon is achieved, the patient is not out of danger unless the underlying inflammatory process is controlled; otherwise, colectomy will still be necessary.
If intensive medical measures do not produce definite improvement within 24 to 48 hours, immediate surgery is required or the patient may die of sepsis caused by bacterial translocation or even perforation.
Maintenance therapy
In general, patients in whom remission was induced using a biologic agent or small molecule should generally continue the same medication for maintenance (1). However, glucocorticoids are tapered based on clinical response and then discontinued, even after effective treatment of a flare, because they are ineffective as maintenance. 5-ASA is more effective in moderate than severe disease, and may be used in conjunction with another agent for maintenance, even if it failed to induce remission initially.
Surgery
Approximately 20 to 40% of patients hospitalized with ulcerative colitis ultimately require surgery (4, 5, 7). Total proctocolectomy is curative: Life expectancy is restored to normal, and the risk of colon cancer is significantly decreased. However, some studies report up to 25% of patients subsequently develop inflammation in the small bowel mucosa consistent with Crohn disease (8, 9); this may occur many years after surgery. After total proctocolectomy with ileal pouch-anal anastomosis (IPAA), there remains a small risk of dysplasia or cancer in the rectal cuff anal transition zone and even in the ileal pouch. After proctocolectomy with ileostomy or IPAA, the quality of life is improved; however, new quality-of-life challenges are created.
Emergency colectomy is indicated for massive hemorrhage, fulminating toxic colitis, or perforation. Subtotal colectomy with ileostomy and rectosigmoid closure (Hartmann procedure) or mucous fistula is usually the procedure of choice because most critically ill patients cannot tolerate more extensive surgery. The rectosigmoid stump may be electively removed later or may be used for ileoanal anastomosis with a pouch. The intact rectal stump should not be allowed to remain indefinitely because of the risks of disease activation and malignant transformation.
Elective surgery is indicated for cancer, symptomatic strictures, growth retardation in children, or, most commonly, intractable chronic disease resulting in severe disability or glucocorticoid dependence. Severe colitis-related extraintestinal manifestations (eg, pyoderma gangrenosum), now better controlled by intensive medical therapies, are only rarely indications for surgery.
The elective procedure of choice in patients with normal sphincter function is restorative proctocolectomy with ileoanal anastomosis. This procedure creates a pelvic reservoir or pouch from distal ileum, which is connected to the anus. The intact sphincter allows continence, typically with 4 to 9 bowel movements/day (including 1 or 2 at night).
Pouchitis is an inflammatory reaction occurring after restorative proctocolectomy with IPAA in approximately 50% of patients within 2 years. The risk of pouchitis appears to be higher in patients with primary sclerosing cholangitis, in patients with preoperative extraintestinal manifestations, and possibly in patients with high preoperative serologic titers of perinuclear antineutrophilic antibodies and other inflammatory bowel disease biomarkers. Pouchitis is thought to be related to bacterial overgrowth. Probiotics, but not antibiotics, are recommended for the prevention of pouchitis (10). Pouchitis is treated with antibiotics (eg, quinolones or metronidazole), or with immunomodulators if refractory to antibiotic therapy (). Pouchitis is treated with antibiotics (eg, quinolones or metronidazole), or with immunomodulators if refractory to antibiotic therapy (11). However, most cases of pouchitis are readily controlled, but some prove refractory to all medical therapy and require conversion to a conventional (Brooke) ileostomy. For patients who prefer management of an ileostomy to the consequences of potential pouchitis, or those who have poor sphincter tone or cannot tolerate frequent bowel movements, the Brooke ileostomy remains an option.
Ileitis after IPAA or end ileostomy is managed the same as other forms of IBD. Surgical procedures are rarely required.
In any event, the physical and emotional burdens imposed by any form of colon resection must be recognized, and care should be taken to see that the patient receives all the instructions and all the medical and psychological support that is necessary before and after surgery.
Treatment references
1. Rubin DT, Ananthakrishnan AN, Siegel CA, Barnes EL, Long MD. ACG Clinical Guideline Update: Ulcerative Colitis in Adults. Am J Gastroenterol. 2025;120(6):1187-1224. Published 2025 Jun 3. doi:10.14309/ajg.0000000000003463
2. Caldera F, Kane S, Long M, Hash JG. AGA Clinical Practice Update on Noncolorectal Cancer Screening and Vaccinations in Patients With Inflammatory Bowel Disease: Expert Review. Clin Gastroenterol Hepatol. 2025;23(5):695-706. doi:10.1016/j.cgh.2024.12.011
3. Ko CW, Singh S, Feuerstein JD, et al. AGA Clinical Practice Guidelines on the Management of Mild-to-Moderate Ulcerative Colitis. Gastroenterology. 2019;156(3):748-764. doi:10.1053/j.gastro.2018.12.009
4. Gros B, Kaplan GG. Ulcerative Colitis in Adults: A Review. JAMA. 2023;330(10):951-965. doi:10.1001/jama.2023.15389
5. Le Berre C, Honap S, Peyrin-Biroulet L. Ulcerative colitis. Lancet. 2023;402(10401):571-584. doi:10.1016/S0140-6736(23)00966-2
6. Feuerstein JD, Isaacs KL, Schneider Y, et al. AGA Clinical Practice Guidelines on the Management of Moderate to Severe Ulcerative Colitis. Gastroenterology. 2020;158(5):1450-1461. doi:10.1053/j.gastro.2020.01.006
7. Burisch J, Lophaven S, Munkholm P, Langholz E. Surgery, cancer and mortality among patients with ulcerative colitis diagnosed 1962-1987 and followed until 2017 in a Danish population-based inception cohort. Aliment Pharmacol Ther. 2022;55(3):339-349. doi:10.1111/apt.16677
8. Hercun J, Côté-Daigneault J, Lahaie RG, et al. Crohn's disease after proctocolectomy and IPAA for ulcerative colitis. Dis Colon Rectum. 64(2):217–224, 2021. doi: 10.1097/DCR.0000000000001721
9. Shamah S, Schneider J, Korelitz BI. High incidence of recurrent Crohn's disease following colectomy for ulcerative colitis revealed with long follow-up. Dig Dis Sci. 63(2):446–451, 2018. doi: 10.1007/s10620-017-4873-7
10. Barnes EL, Agrawal M, Syal G, et al. AGA Clinical Practice Guideline on the Management of Pouchitis and Inflammatory Pouch Disorders. Gastroenterology. 2024;166(1):59-85. doi:10.1053/j.gastro.2023.10.015
11. Syal G, Barnes E, Raffals L, et al. Medical Therapies for Prevention and Treatment of Inflammatory Pouch Disorders-A Systematic Review and Meta-Analysis. Am J Gastroenterol. 2025;120(1):135-150. doi:10.14309/ajg.0000000000003136
Prognosis for Ulcerative Colitis
Usually, ulcerative colitis is chronic with repeated exacerbations and remissions. In some of patients, an initial attack becomes fulminant with massive hemorrhage, perforation, or sepsis and toxemia. Complete recovery after a single attack occurs in a few patients.
Patients with localized ulcerative proctitis have the best prognosis (1). Severe systemic manifestations, toxic complications, and malignant degeneration are unlikely, and late extension of the disease occurs only in a small group of patients. Surgery is rarely required, and life expectancy is normal. The symptoms, however, may prove stubborn and refractory. Moreover, because extensive ulcerative colitis may begin in the rectum and spread proximally, proctitis should not be considered localized until it has been observed for ≥ 6 months. Localized disease that later extends is often more severe and more refractory to therapy (2).
Colon cancer
The risk of colon cancer is proportional to the duration of disease and amount of colon affected but not necessarily to the clinical severity of the attacks (3). Some studies suggest that sustained microscopic inflammation is a risk factor, and sustained remission appears to be the most important protective factor (4).
The high risk of colon cancer conferred by IBD has been noted in multiple studies. More recent data suggests that the prevalence of colon cancer is decreasing and was overestimated by researches at tertiary care centers (5).
Patients who have inflammatory bowel disease and primary sclerosing cholangitis are at a higher risk of cancer from the time of colitis diagnosis (6).
Regular colonoscopic surveillance, preferably during remission, is advised for patients with disease duration > 8 years (except for those with isolated proctitis) or when there is concomitant primary sclerosing cholangitis, in which case surveillance colonoscopy should begin at the time of diagnosis (6). Expert guidelines also suggest doing random ("nontargeted") biopsies (taken every 10 cm throughout the colon) when using high-definition white-light colonoscopy but doing only targeted biopsies of visible lesions when using chromoendoscopy to detect dysplasia (7, 8). Definite dysplasia of any grade within an area affected by colitis is liable to progress to more advanced neoplasia and even cancer. After complete removal of endoscopically resectable polypoid or nonpolypoid dysplastic lesions, colonoscopic surveillance is suggested rather than colectomy. Patients with dysplasia that is not visible endoscopically should probably be referred to a gastroenterologist with expertise in IBD surveillance using chromoendoscopy and/or high-definition colonoscopy to decide whether colectomy or continued colonoscopic surveillance should be performed.
The optimal frequency of colonoscopic surveillance has not been established, but some authorities recommend every 1 to 5 years based on multiple factors like presence of primary sclerosing cholangitis, family history of colon cancer, prior dysplasia, active inflammation, pseudopolyps, colonic stricture, and disease extent (8, 9).
The 5-year survival after diagnosis of colitis-related cancer is approximately 75%, a figure comparable to that for colorectal cancer in the general population (10, 11).
Prognosis references
1. Fumery M, Singh S, Dulai PS, Gower-Rousseau C, Peyrin-Biroulet L, Sandborn WJ. Natural History of Adult Ulcerative Colitis in Population-based Cohorts: A Systematic Review. Clin Gastroenterol Hepatol. 2018;16(3):343-356.e3. doi:10.1016/j.cgh.2017.06.016
2. Roda G, Narula N, Pinotti R, et al. Systematic review with meta-analysis: proximal disease extension in limited ulcerative colitis. Aliment Pharmacol Ther. 2017;45(12):1481-1492. doi:10.1111/apt.14063
3. Rex DK, Anderson JC, Butterly LF, et al. Quality Indicators for Colonoscopy. Am J Gastroenterol. 2024;119(9):1754-1780. doi:10.14309/ajg.0000000000002972
4. Singh S, Loftus EV Jr, Limketkai BN, et al. AGA Living Clinical Practice Guideline on Pharmacological Management of Moderate-to-Severe Ulcerative Colitis. Gastroenterology. 2024;167(7):1307-1343. doi:10.1053/j.gastro.2024.10.001
5. Stidham RW, Higgins PDR. Colorectal cancer in inflammatory bowel disease. Clin Colon Rectal Surg. 31(3):168-178, 2018. doi: 10.1055/s-0037-1602237
6. Rubin DT, Ananthakrishnan AN, Siegel CA, Sauer BG, Long MD. ACG Clinical Guideline: Ulcerative Colitis in Adults. Am J Gastroenterol. 2019;114(3):384-413. doi:10.14309/ajg.0000000000000152
7. National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines). Colorectal Cancer Screening, version 2.2025–June 24, 2025. https://www.nccn.org/professionals/physician_gls/pdf/colorectal_screening.pdf. Accessed February27, 2026.
8. Murthy SK, Feuerstein JD, Nguyen GC, Velayos FS. AGA Clinical Practice Update on Endoscopic Surveillance and Management of Colorectal Dysplasia in Inflammatory Bowel Diseases: Expert Review. Gastroenterology. 2021;161(3):1043-1051.e4. doi:10.1053/j.gastro.2021.05.063
9. Laine L, Kaltenbach T, Barkun A, et al. SCENIC international consensus statement on surveillance and management of dysplasia in inflammatory bowel disease. Gastroenterology. 148(3):639-651.e28, 2015. doi: 10.1053/j.gastro.2015.01.031
10. Raje P, Sonal S, Qwaider YZ, et al. Colitis-Associated Colorectal Cancer Survival is Comparable to Sporadic Cases after Surgery: a Matched-Pair Analysis. J Gastrointest Surg. 2023;27(7):1423-1428. doi:10.1007/s11605-023-05692-y
11. Sugita A, Ikeuchi H, Funayama Y, et al. Postoperative Survival in Colitis-associated Colorectal Cancer With Ulcerative Colitis in Japan: A Multicenter Analysis. Anticancer Res. 2021;41(5):2681-2688. doi:10.21873/anticanres.15049
Key Points
Ulcerative colitis begins in the rectum and may extend proximally in a contiguous fashion without intervening patches of normal bowel.
Symptoms are intermittent episodes of abdominal cramping and bloody diarrhea.
Complications include fulminant colitis, which may lead to perforation; long-term, the risk of colon cancer is increased.
Treat mild disease with 5-ASA by rectum and, for proximal disease, by mouth.
Treat extensive or severe disease with high-dose glucocorticoids, immunomodulator therapy (eg, azathioprine, 6-mercaptopurine), biologics (eg, adalimumab, infliximab, vedolizumab), tofacitinib, upadacitinib, or ozanimod.Treat extensive or severe disease with high-dose glucocorticoids, immunomodulator therapy (eg, azathioprine, 6-mercaptopurine), biologics (eg, adalimumab, infliximab, vedolizumab), tofacitinib, upadacitinib, or ozanimod.
Treat fulminant disease with high-dose IV glucocorticoids or cyclosporine and antibiotics (eg, metronidazole, ciprofloxacin) or infliximab; colectomy may be required.Treat fulminant disease with high-dose IV glucocorticoids or cyclosporine and antibiotics (eg, metronidazole, ciprofloxacin) or infliximab; colectomy may be required.
Up to 40% of patients with extensive ulcerative colitis ultimately require surgery.